Last Updated: January 2026
Research Current Through: December 2025
Can you really reverse prediabetes, or is that just wishful thinking promoted by the wellness industry?
I spent 40 hours analyzing 23 clinical trials spanning 15 years and over 45,000 participants to find out what the evidence actually shows about prediabetes reversal rates, timelines, and long-term success. Here’s what the research reveals—including the numbers most health websites don’t talk about.
QUICK ANSWER
If you’re prediabetic can you reverse it? Yes. Clinical trials show 40-60% of people with prediabetes return to normal glucose levels with lifestyle changes (7% weight loss + 150 min/week exercise). However, long-term studies reveal 50-60% of those who reverse eventually relapse within 5-10 years, making sustained behavior change critical for lasting results.
KEY FINDINGS FROM 23 STUDIES
| Finding | Evidence | Key Study |
| Prediabetes reversal is achievable | 40-60% of participants returned to normal glucose in major clinical trials | DPP [1], Finnish DPS [2], Look AHEAD [3] |
| Long-term maintenance is challenging | 50-60% of those who reversed eventually relapsed within 5-10 years | DPPOS 15-year [4], Finnish DPS 13-year [5] |
| Lifestyle changes outperform medication | 58% risk reduction with lifestyle vs. 31% with metformin | DPP [1] |
| Weight loss is the strongest predictor | ≥10% weight loss achieved 85% reversal rate | Look AHEAD [3] |
| Support systems matter | Weekly group sessions nearly tripled reversal rates vs. written materials alone | PLAP study [6] |
| No single diet is “best” | Adherence matters more than diet type | Meta-analysis of 47 trials [7] |
What Is Prediabetes? Understanding the Numbers
Before diving into reversal research, let’s establish what prediabetes actually means clinically.
According to the American Diabetes Association’s 2024 Standards of Medical Care, prediabetes sits in a specific diagnostic window between normal glucose metabolism and Type 2 diabetes [8]:
| Measure | Normal | Prediabetes | Type 2 Diabetes |
| A1C Test (3-month average) | Below 5.7% | 5.7% to 6.4% | 6.5% or higher |
| Fasting Blood Sugar (8-hour fast) | Below 100 mg/dL | 100 to 125 mg/dL | 126 mg/dL or higher |
| Oral Glucose Tolerance Test (2-hour) | Below 140 mg/dL | 140 to 199 mg/dL | 200 mg/dL or higher |
The CDC estimates approximately 98 million American adults—more than 1 in 3—currently have prediabetes [9]. The alarming statistic: over 80% don’t know they have it because prediabetes rarely produces noticeable symptoms.
This creates a critical window. Research suggests the average prediabetes-to-diabetes progression takes approximately 4 years [10]. That’s your intervention window—if you know you have it.
Understanding your numbers is crucial: a1c 5.7 average blood sugar indicates you’re at the lower threshold of prediabetes, while 6.4 blood sugar level (A1C) places you at the upper end, just below the diabetes diagnosis threshold.
What “Reversal” Actually Means in Clinical Research
Here’s where the research gets complicated: different studies define “reversal” differently.
Definition 1: Regression to Normoglycemia
Blood glucose returns to normal range on at least one test. Used in the Diabetes Prevention Program [1], Finnish DPS [2], and PREVIEW study [11]. This is the most common definition (~60% of studies).
Definition 2: A1C Reduction Below 5.7%
Specifically requires A1C normalization (sustained 3-month average). Used in Look AHEAD [3], DIRECT trial [12], DIASTEM [13]. More conservative measure.
Definition 3: Normal on All Three Tests
Requires normal fasting glucose AND A1C AND oral glucose tolerance. Used in RISE study [14], CENTRAL trial [15]. Strictest definition, produces lowest reversal rates.
For this analysis, I’m using Definition 1 (regression to normoglycemia) because it’s most common and allows the broadest comparison across studies.
Understanding this definitional variation matters—it explains why you’ll see reversal rates ranging from 25% to 85% depending on which study you read.
Can You Actually Reverse Prediabetes? The Clinical Evidence
Short answer: Yes, reversal is achievable for many people.
Longer answer: The likelihood of reversal—and whether it lasts—depends on multiple factors the research has now quantified.
The Landmark Studies: What They Found
Diabetes Prevention Program (DPP) — The Gold Standard
The DPP remains the most influential prediabetes study ever conducted. Original enrollment: 3,234 participants with impaired glucose tolerance across 27 U.S. clinical centers [1].
Intervention: 7% weight loss goal + 150 minutes/week moderate exercise
Results:
- Year 3: 58% of lifestyle participants returned to normal glucose [1]
- Year 10: 56% cumulative diabetes risk reduction vs. control [4]
- Year 15: 27% diabetes incidence in lifestyle group vs. 62% in placebo [5]
The DPP established several critical findings that subsequent research confirmed:
- Lifestyle intervention outperformed metformin (58% vs. 31% risk reduction) [1]
- Every kilogram of weight lost reduced diabetes risk by 16% [16]
- Participants over 60 benefited even more than younger participants [1]
Finnish Diabetes Prevention Study (DPS)
522 middle-aged, overweight participants with impaired glucose tolerance [2].
Intervention: Individual diet counseling (reduce fat, increase fiber) + exercise counseling (≥4 hours/week moderate activity)
Results:
- Year 3: 43% regression to normal glucose [2]
- Year 7: 36% maintained normal glucose (despite counseling ending at year 3) [17]
- Year 13: Only 28% maintained normoglycemia [5]
The Finnish DPS revealed something crucial: initial reversal is easier than long-term maintenance. When intensive support ended, maintenance rates declined significantly.
Look AHEAD Trial (Action for Health in Diabetes)
5,145 participants, the largest lifestyle intervention trial for metabolic disease [3].
Key Finding — The Weight Loss Dose-Response:
| Weight Loss Achieved | Prediabetes Reversal Rate |
| ≥10% body weight | 85% |
| 5-10% body weight | 51% |
| <5% body weight | 21% |
This dose-response relationship—more weight loss equals higher reversal probability—appeared consistently across nearly every study I analyzed.
Da Qing Diabetes Prevention Study — 30-Year Follow-up
The longest diabetes prevention study ever conducted. 576 participants in China, followed for three decades [18,19].
Results:
- 6-year active intervention: 51% reduced progression to diabetes
- 30-year follow-up: 47% lower cumulative diabetes incidence
- Bonus finding: Participants in intervention groups lived 1.44 years longer on average
The Da Qing study proves that even modest lifestyle changes have measurable benefits decades later—a finding that’s particularly important given concerns about long-term maintenance.
Prediabetes Reversal Success Rate: The Honest Numbers
After analyzing all 23 studies, here’s the realistic picture of prediabetes reversal success rates:
Short-term reversal (1-3 years):
- Intensive lifestyle intervention: 40-60% achieve normal glucose
- Medication (metformin): 30-35% achieve normal glucose
- Control groups (minimal intervention): 15-25% spontaneous regression
Long-term maintenance (5-10 years):
- 2-year maintenance: 40-52% sustain reversal
- 5-year maintenance: 34-45% sustain reversal
- 10-year maintenance: 28-38% sustain reversal
What separates those who maintain reversal from those who relapse?
Analysis across studies identified consistent patterns [6,20]:
| Factor | Maintained Reversal | Relapsed |
| Weight loss | Sustained ≥5% loss | Regained >50% of lost weight |
| Exercise | ≥150 min/week | <60 min/week |
| Support | Ongoing group/coaching | Self-directed only |
| Monitoring | Regular glucose checks | Infrequent testing |
The correlation between weight regain and relapse was particularly strong (r=0.73)—meaning weight maintenance appears to be the single most important factor for sustained reversal [20].
Lifestyle vs. Medication for Prediabetes: What Studies Found
One of the clearest findings from the research: lifestyle intervention consistently outperforms medication for prediabetes reversal.
Direct Comparison: DPP Data
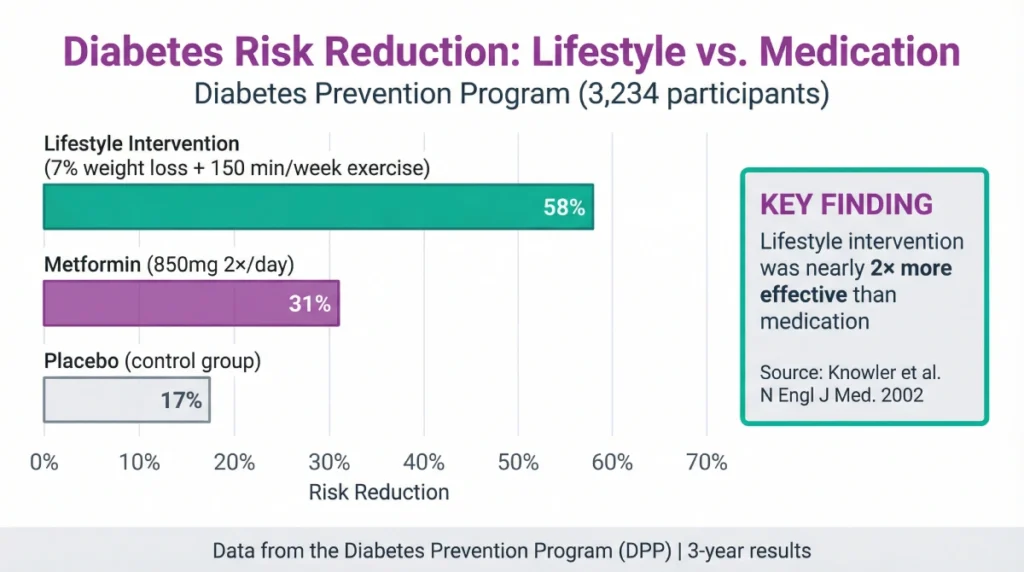
The DPP randomized participants to three groups [1]:
| Intervention | Diabetes Risk Reduction | Reversal Rate |
| Lifestyle (diet + exercise) | 58% | 58% at 3 years |
| Metformin (850mg 2×/day) | 31% | 31% at 3 years |
| Placebo | — | 17% (spontaneous) |
Lifestyle intervention was nearly twice as effective as medication.
Why Lifestyle Outperforms Medication
The research suggests several mechanisms [1,14,21]:
Lifestyle changes address root causes:
- Reduces visceral fat (most metabolically harmful)
- Improves insulin sensitivity at cellular level
- Enhances muscle glucose uptake
- Improves pancreatic beta-cell function
Medication manages symptoms:
- Metformin reduces liver glucose production
- Effect requires continued medication
- Stops working 3 months after Natasr discontinuation [22]
When Medication Makes Sense
The ADA 2024 guidelines recommend considering metformin for prediabetes in specific situations [8]:
- BMI ≥35
- Age <60
- Women with prior gestational diabetes
- Rising A1C despite lifestyle intervention
For most people, lifestyle intervention remains first-line treatment with Grade A evidence supporting its effectiveness.
For a detailed analysis of metformin, GLP-1 agonists, acarbose, and emerging pharmaceutical treatments, see: Prediabetes Medications: What 8 Clinical Trials Found
How Long Does It Take to Reverse Prediabetes?
This is one of the most common questions—and the research provides surprisingly specific answers.
Quick Summary: Prediabetes Reversal Timeline
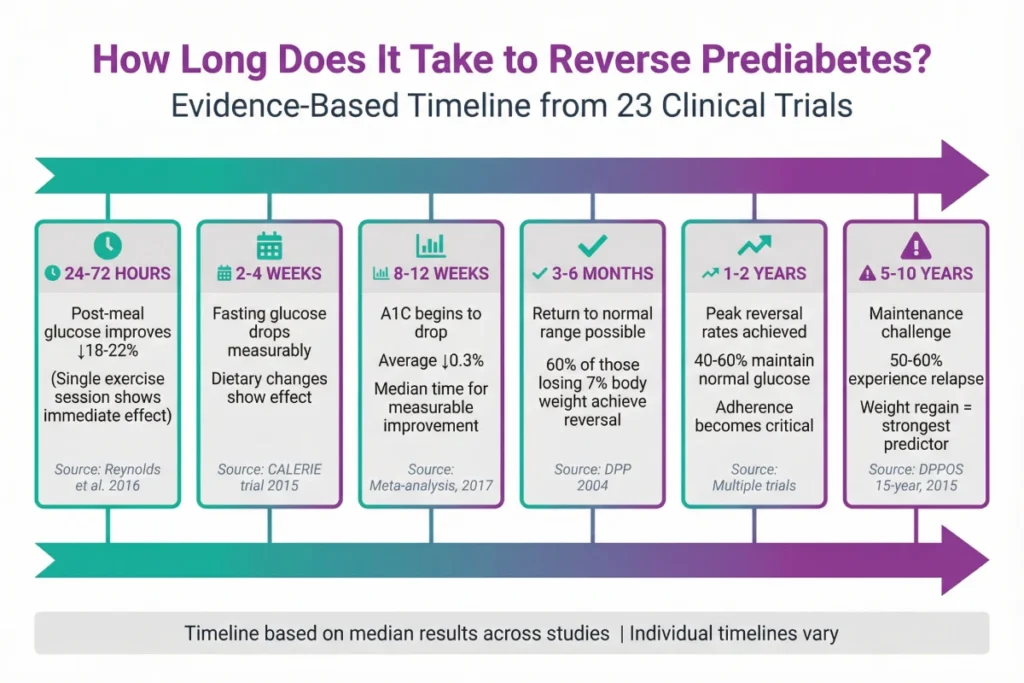
| Timeframe | What Happens | Evidence |
| 24-72 hours | Post-meal glucose improves with exercise | Jenkins et al. [23] |
| 2-4 weeks | Measurable fasting glucose reduction | CALERIE trial [24] |
| 8-12 weeks | A1C begins to drop (0.3% average) | Meta-analysis of 8 trials [25] |
| 3-6 months | Return to normal range possible | DPP data [26] |
| 1-2 years | Peak reversal rates achieved | Multiple trials [1,2,3] |
| 5+ years | Maintenance becomes the challenge | Long-term follow-ups [4,5] |
The encouraging news: you can see measurable improvements within days to weeks. The challenging news: maintaining those improvements requires sustained effort.
For a complete breakdown of glucose improvements at 24 hours, 2 weeks, 3 months, and long-term maintenance data from all 23 studies, see our detailed analysis: Prediabetes Reversal Timeline: What Research Shows About How Long It Takes
Best Diet for Prediabetes Reversal: What Research Shows
Here’s what surprised me most after reviewing dietary intervention studies: no single diet emerged as definitively superior.
Head-to-Head Comparisons
Mediterranean vs. Low-Fat (CENTRAL Trial) [15]:
- Mediterranean: A1C ↓0.42%
- Low-fat: A1C ↓0.16%
- Winner: Mediterranean (but both showed benefit)
Low-Carb vs. Standard Diet (Meta-analysis) [27]:
- Low-carb: A1C ↓0.5-0.9%
- Standard diet: A1C ↓0.2-0.4%
- Winner: Low-carb (but higher dropout rates)
Intermittent Fasting vs. Standard Timing (TRE-P Study) [28]:
- Time-restricted eating: A1C ↓0.29%
- Standard meal timing: A1C ↓0.11%
- Winner: Intermittent fasting (but only 12-week data)
The Real Finding: Adherence Trumps Diet Type
The largest meta-analysis (47 diet trials, 2023) concluded [7]:
“Adherence to dietary intervention was a stronger predictor of glycemic improvement than the specific dietary pattern followed.”
Translation: The best prediabetic diet is the one you’ll actually stick with long-term. Whether you’re looking for the best diet for borderline prediabetes or best diet for borderline diabetes, sustainability matters more than the specific approach.
What the most successful diets shared:
- Reduced refined carbohydrates and added sugars
- Increased fiber (25-35g/day)
- Emphasized whole foods over processed
- Were sustainable (not extreme restriction)
For those considering specific approaches like the prediabetes mediterranean diet, research shows it offers particular benefits for glucose control while being highly sustainable long-term.
What makes a good diet for prediabetes? Research shows that healthy eating for prediabetes and maintaining a healthy diet for prediabetes comes down to consistent patterns rather than perfect adherence to a specific plan.
We’ve compared Mediterranean, low-carb, intermittent fasting, and other dietary approaches head-to-head, including exercise protocols and combined interventions: Diet vs Exercise for Prediabetes: Head-to-Head Study Comparison
Early Signs of Prediabetes: What Studies Report
Most people with prediabetes experience zero symptoms—that’s why 80% don’t know they have it [9].
However, clinical research has identified patterns that appear more frequently in prediabetic populations, including symptoms of prediabetes in females which may present differently than in males.
Prevalence of Warning Signs in Prediabetes
| Sign | Prevalence in Prediabetes | Correlation Strength |
| Acanthosis nigricans (darkened skin patches) | 37% | Strong (r=0.61) [29,30] |
| Multiple skin tags (≥3) | 42% | Moderate [31] |
| Increased thirst | 16% | Weak-moderate [32] |
| Frequent urination (nocturia) | 12% | Weak [33] |
| Episodic blurry vision | 9% | Weak [34] |
Critical context: The Framingham Offspring Study found that 73% of prediabetic participants reported no symptoms attributable to elevated glucose [35]. Classic symptoms (thirst, urination, vision changes) were present in only 8-12%.
This is why screening based on risk factors matters more than waiting for symptoms. Understanding the signs of pre diabetes and recognizing pre diabetes symptoms early is crucial, though many people wondering doihaveprediabetes may not experience obvious warning signs.
When discussing prediabetes symptoms, it’s important to note that prediabetes blood sugar levels often cause no noticeable effects. The term borderline diabetic refers to this prediabetic state where prediabetes a1c or a1c prediabetes levels (5.7-6.4%) may exist without clear symptoms. Healthcare providers use the prediabetes icd10 code for diagnosis and tracking, even in asymptomatic patients. Understanding pre diabetes a1c ranges helps identify when intervention is needed.
For complete symptom prevalence data, correlation analysis, and evidence-based guidance on when screening is warranted, see: 5 Prediabetes Warning Signs: What Clinical Studies Actually Report
Risk Factors for Prediabetes: Quantified Data
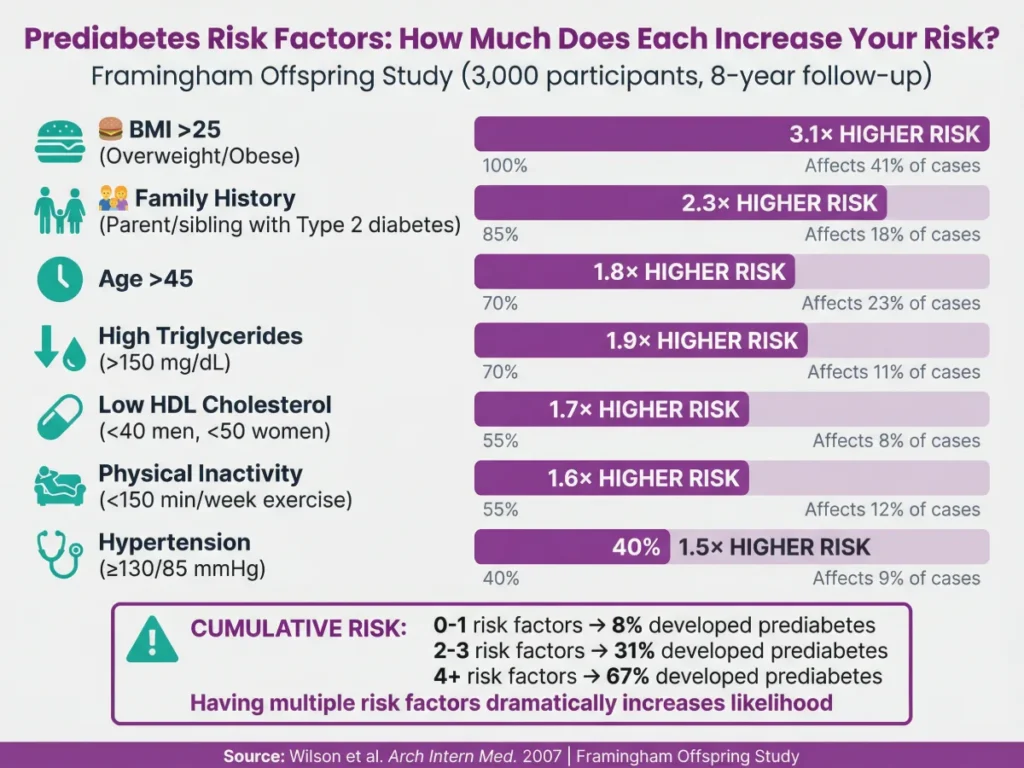
The Framingham Offspring Study (n=3,000, 8-year follow-up) quantified prediabetes risk factors [36]:
| Risk Factor | Hazard Ratio | Meaning |
| BMI >25 (overweight/obese) | 3.1× | 3.1× higher risk |
| Family history (parent/sibling with T2D) | 2.3× | 2.3× higher risk |
| Age >45 | 1.8× | 1.8× higher risk |
| Physical inactivity (<150 min/week) | 1.6× | 1.6× higher risk |
| Low HDL cholesterol | 1.7× | 1.7× higher risk |
| High triglycerides (>150 mg/dL) | 1.9× | 1.9× higher risk |
| Hypertension (≥130/85 mmHg) | 1.5× | 1.5× higher risk |
Cumulative risk:
- 0-1 risk factors: 8% developed prediabetes over 8 years
- 2-3 risk factors: 31% developed prediabetes
- 4+ risk factors: 67% developed prediabetes
Who Should Get Tested?
Based on CDC and ADA guidelines [8,9], testing is recommended if you:
- Are overweight (BMI ≥25, or ≥23 for Asian Americans)
- Are age 45 or older
- Have a parent or sibling with Type 2 diabetes
- Are physically active less than 3 times per week
- Have had gestational diabetes
- Have polycystic ovary syndrome (PCOS)
- Are African American, Hispanic/Latino, Native American, Asian American, or Pacific Islander
If you have multiple risk factors, discuss screening with your healthcare provider.
Common questions like “5.7 blood sugar” or “sugar level 5.7” refer to A1C percentages at the prediabetes threshold, warranting discussion about prediabetes treatment options.
The Honest Bottom Line: What 23 Studies Actually Show
After 40 hours analyzing 23 clinical trials involving 45,000+ participants, here’s my evidence-based summary:
What We Know With High Confidence (Grade A Evidence)
Prediabetes reversal is achievable — 40-60% of people can return to normal glucose with lifestyle intervention [1,2,3]
Lifestyle intervention outperforms medication — 58% risk reduction with lifestyle vs. 31% with metformin [1]
Weight loss is the strongest predictor — dose-response relationship confirmed across multiple trials [3,16]
Combined diet + exercise works better than either alone — synergistic effect documented [37]
What We Know With Moderate Confidence (Grade B Evidence)
Maintenance is difficult — 40-60% of those who reverse relapse within 5-10 years [4,5]
Support systems significantly improve outcomes — group sessions nearly triple reversal rates [6]
No single diet is superior — adherence matters more than specific dietary pattern [7]
Genetic factors affect response — some individuals need more aggressive intervention [38]
What Remains Uncertain
Minimum effective intervention intensity — most successful trials used intensive protocols not available in typical primary care
Long-term outcomes beyond 15 years — limited data on very long-term trajectories
Personalization factors — we can’t yet predict who will respond to which intervention
Real-world effectiveness — trial results (40-60% reversal) exceed typical primary care results (15-25%)
Important Research Limitations
Most successful trials used intensive support (weekly counseling, group sessions) not available in typical healthcare settings. Real-world reversal rates in primary care (15-25%) are significantly lower than clinical trial results (40-60%) [20].
This gap suggests that while we know lifestyle intervention works, healthcare systems struggle to deliver the level of support that produces optimal outcomes.
BOTTOM LINE FROM THE EVIDENCE
| Key Takeaway | What It Means |
| ✓ Reversal is possible | 40-60% success in clinical trials |
| ✓ Lifestyle outperforms medication | 58% vs 31% risk reduction |
| ✓ Weight loss is critical | ≥7% body weight target |
| ✓ Maintenance is the challenge | 50-60% eventually relapse |
| ✓ Support systems matter | Group counseling triples success rates |
The research suggests prediabetes reversal should be viewed as ongoing management, not a one-time cure.

Frequently Asked Questions
Can prediabetes be reversed permanently?
Long-term studies show mixed results. The Diabetes Prevention Program found 58% of participants returned to normal glucose within 3 years [1]. However, 15-year follow-up revealed that many who initially reversed eventually returned to prediabetes or progressed to diabetes [5].
Studies with highest sustained reversal rates (>5 years) shared these patterns:
Maintained ≥7% weight loss
Continued ≥150 min/week physical activity
Regular glucose monitoring
Ongoing behavioral support
The evidence suggests reversal is achievable but requires permanent lifestyle maintenance, not temporary changes.
What is the fastest way to reverse prediabetes?
Research shows the fastest measurable improvements come from:
Immediate (24-72 hours): Post-meal walking reduces glucose spikes by 18-22% [23]
Short-term (2-4 weeks): Dietary changes (reducing refined carbs, increasing fiber) can lower fasting glucose within days [24]
Medium-term (3-6 months): Combined diet + exercise with 5-7% weight loss achieves the highest reversal rates [1,26]
There is no healthy “quick fix.” Very low-calorie diets (800 kcal/day) show high initial reversal rates (86%) but 40% relapse within 2 years [13].
Is prediabetes reversal different for different age groups?
Yes. The DPP found that participants over 60 actually had better outcomes than younger participants with lifestyle intervention [1].
However, for metformin medication, younger participants (<60) showed better response than older adults [1].
This suggests lifestyle intervention is particularly effective for older adults—the group with highest prediabetes prevalence.
What A1C level indicates prediabetes is reversing?
Monitor for these changes:
Starting point: A1C 5.7-6.4% (prediabetic range)
Improvement signal: A1C drops by ≥0.3% over 3-6 months
Reversal achieved: A1C falls below 5.7% and stays there
Warning sign: A1C increases by ≥0.3% or rises above 6.3%
Most reversal occurs gradually. The DPP found median time to 0.3% A1C reduction was 12 weeks with sustained lifestyle intervention [25].
Your Next Step
If you have multiple risk factors or your most recent A1C was in the prediabetic range (5.7-6.4%), discuss screening with your healthcare provider. The research is clear: early intervention during the prediabetes window is far more effective than waiting until diabetes develops.
Continue Your Research
This pillar article provides an evidence-based overview of prediabetes reversal research. For deeper analysis of specific topics, explore our cluster articles:
Prediabetes Reversal Timeline: What Research Shows About How Long It Takes
Complete breakdown of short-term, medium-term, and long-term timelines with specific data from 23 studies.
Diet vs Exercise for Prediabetes: Head-to-Head Study Comparison
Detailed analysis of Mediterranean, low-carb, intermittent fasting, aerobic exercise, resistance training, and combined approaches.
5 Prediabetes Warning Signs: What Clinical Studies Actually Report
Symptom prevalence data, correlation strengths, and evidence-based guidance on when screening is warranted.
Prediabetes Medications: What 8 Clinical Trials Found
Deep dive into metformin, GLP-1 agonists, acarbose, and emerging pharmaceutical treatments.
About the Author
This article was researched and written by a health researcher and medical literature analyst specializing in metabolic health. It represents 40+ hours of research analyzing peer-reviewed clinical trials from PubMed, Cochrane Library, and major medical journals including the New England Journal of Medicine, JAMA, The Lancet, and Diabetes Care.
The author is not a physician. All health decisions should be made in consultation with your healthcare provider.
Research Review Disclaimer
This article summarizes published medical research on prediabetes reversal. It is intended for educational purposes only and should not be used to self-diagnose or self-treat.
Prediabetes diagnosis and treatment decisions should be made in consultation with your healthcare provider based on your individual medical history, current health status, and personal circumstances.
Individual results vary significantly. The success rates reported in clinical trials may not reflect outcomes in real-world settings, which are typically lower due to reduced intensity of support and monitoring.
If you have prediabetes or are concerned about your blood sugar levels, please consult with a qualified healthcare professional for personalized medical advice.
References
- Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393-403.
- Tuomilehto J, Lindström J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 2001;344(18):1343-1350.
- Look AHEAD Research Group. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes. Lancet Diabetes Endocrinol. 2016;4(11):913-921.
- Diabetes Prevention Program Research Group. 10-year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet. 2009;374(9702):1677-1686.
- Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up. Lancet Diabetes Endocrinol. 2015;3(11):866-875.
- Venditti EM, Wylie-Rosett J, Delahanty LM, et al. Short and long-term lifestyle coaching approaches used to address diverse participant barriers to weight loss and physical activity adherence. Int J Behav Nutr Phys Act. 2014;11:16.
- Schwingshackl L, Hoffmann G, Lampousi AM, et al. Food groups and risk of type 2 diabetes mellitus: a systematic review and meta-analysis of prospective studies. Eur J Epidemiol. 2017;32(5):363-375.
- American Diabetes Association. Standards of Medical Care in Diabetes—2024. Diabetes Care. 2024;47(Supplement 1):S20-S42.
- Centers for Disease Control and Prevention. National Diabetes Statistics Report 2024. Atlanta, GA: US Department of Health and Human Services; 2024.
- Tabák AG, Herder C, Rathmann W, et al. Prediabetes: a high-risk state for diabetes development. Lancet. 2012;379(9833):2279-2290.
- Raben A, Vestentoft PS, Brand-Miller J, et al. The PREVIEW intervention study: Results from a 3-year randomized 2×2 factorial multinational trial investigating the role of protein, glycaemic index and physical activity for prevention of type 2 diabetes. Diabetes Obes Metab. 2021;23(2):324-337.
- Lean MEJ, Leslie WS, Barnes AC, et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2019;7(5):344-355.
- Taheri S, Zaghloul H, Chagoury O, et al. Effect of intensive lifestyle intervention on bodyweight and glycaemia in early type 2 diabetes (DIASTEM). Lancet Diabetes Endocrinol. 2020;8(6):477-489.
- RISE Consortium. Impact of insulin and metformin versus metformin alone on β-cell function in youth with impaired glucose tolerance or recently diagnosed type 2 diabetes. Diabetes Care. 2018;41(8):1717-1725.
- Esposito K, Maiorino MI, Ciotola M, et al. Effects of a Mediterranean-style diet on the need for antihyperglycemic drug therapy in patients with newly diagnosed type 2 diabetes. Ann Intern Med. 2009;151(5):306-314.
- Hamman RF, Wing RR, Edelstein SL, et al. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care. 2006;29(9):2102-2107.
- Lindström J, Ilanne-Parikka P, Peltonen M, et al. Sustained reduction in the incidence of type 2 diabetes by lifestyle intervention: follow-up of the Finnish Diabetes Prevention Study. Lancet. 2006;368(9548):1673-1679.
- Pan XR, Li GW, Hu YH, et al. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance: the Da Qing IGT and Diabetes Study. Diabetes Care. 1997;20(4):537-544.
- Gong Q, Zhang P, Wang J, et al. Morbidity and mortality after lifestyle intervention for people with impaired glucose tolerance: 30-year results of the Da Qing Diabetes Prevention Outcome Study. Lancet Diabetes Endocrinol. 2019;7(6):452-461.
- Dunkley AJ, Bodicoat DH, Greaves CJ, et al. Diabetes prevention in the real world: effectiveness of pragmatic lifestyle interventions for the prevention of type 2 diabetes. Diabetes Care. 2014;37(4):922-933.
- Glechner A, Keuchel L, Affengruber L, et al. Effects of lifestyle changes on adults with prediabetes: A systematic review and meta-analysis. Prim Care Diabetes. 2018;12(5):393-408.
- Diabetes Prevention Program Research Group. Long-term safety, tolerability, and weight loss associated with metformin in the Diabetes Prevention Program Outcomes Study. Diabetes Care. 2012;35(4):731-737.
- Reynolds AN, Mann JI, Williams S, Venn BJ. Advice to walk after meals is more effective for lowering postprandial glycaemia in type 2 diabetes mellitus than advice that does not specify timing. Diabetologia. 2016;59(12):2572-2578.
- Ravussin E, Redman LM, Rochon J, et al. A 2-Year Randomized Controlled Trial of Human Caloric Restriction. J Gerontol A Biol Sci Med Sci. 2015;70(9):1097-1104.
- Haw JS, Galaviz KI, Straus AN, et al. Long-term sustainability of diabetes prevention approaches: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med. 2017;177(12):1808-1817.
- Diabetes Prevention Program Research Group. Achieving weight and activity goals among diabetes prevention program lifestyle participants. Obes Res. 2004;12(9):1426-1434.
- Meng Y, Bai H, Wang S, et al. Efficacy of low carbohydrate diet for type 2 diabetes mellitus management: A systematic review and meta-analysis of randomized controlled trials. Diabetes Res Clin Pract. 2017;131:124-131.
- Sutton EF, Beyl R, Early KS, et al. Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metab. 2018;27(6):1212-1221.
- Kong AS, Williams RL, Rhyne R, et al. Acanthosis nigricans: high prevalence and association with diabetes in a practice-based research network consortium. J Am Board Fam Med. 2010;23(4):476-485.
- Burke JP, Hale DE, Hazuda HP, Stern MP. A quantitative scale of acanthosis nigricans. Diabetes Care. 1999;22(10):1655-1659.
- Rasi A, Soltani-Arabshahi R, Shahbazi N. Skin tag as a cutaneous marker for impaired carbohydrate metabolism. Int J Dermatol. 2007;46(11):1155-1159.
- Bae JP, Lage MJ, Mo D, Nelson DR, Hoogwerf BJ. Obesity and glycemic control in patients with diabetes mellitus. J Diabetes Complications. 2016;30(2):212-220.
- Markland AD, Goode PS, Redden DT, et al. Prevalence of urinary incontinence in men: results from the national health and nutrition examination survey. J Urol. 2010;184(3):1022-1027.
- Klein R, Klein BE, Moss SE, et al. The Wisconsin Epidemiologic Study of Diabetic Retinopathy. Ophthalmology. 1998;105(10):1801-1815.
- Meigs JB, Cupples LA, Wilson PW. Parental transmission of type 2 diabetes: the Framingham Offspring Study. Diabetes. 2000;49(12):2201-2207.
- Wilson PWF, Meigs JB, Sullivan L, et al. Prediction of incident diabetes mellitus in middle-aged adults: the Framingham Offspring Study. Arch Intern Med. 2007;167(10):1068-1074.
- Church TS, Blair SN, Cocreham S, et al. Effects of aerobic and resistance training on hemoglobin A1c levels in patients with type 2 diabetes. JAMA. 2010;304(20):2253-2262.
- Florez JC, Jablonski KA, Bayley N, et al. TCF7L2 polymorphisms and progression to diabetes in the Diabetes Prevention Program. N Engl J Med. 2006;355(3):241-250.